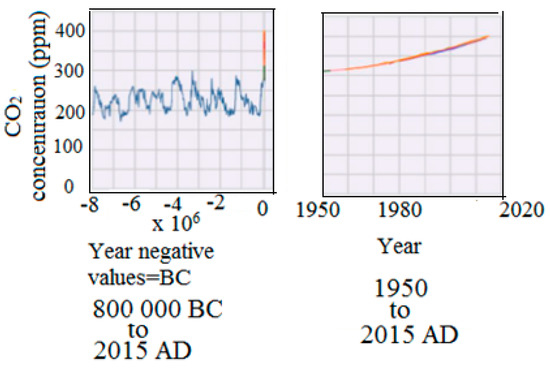
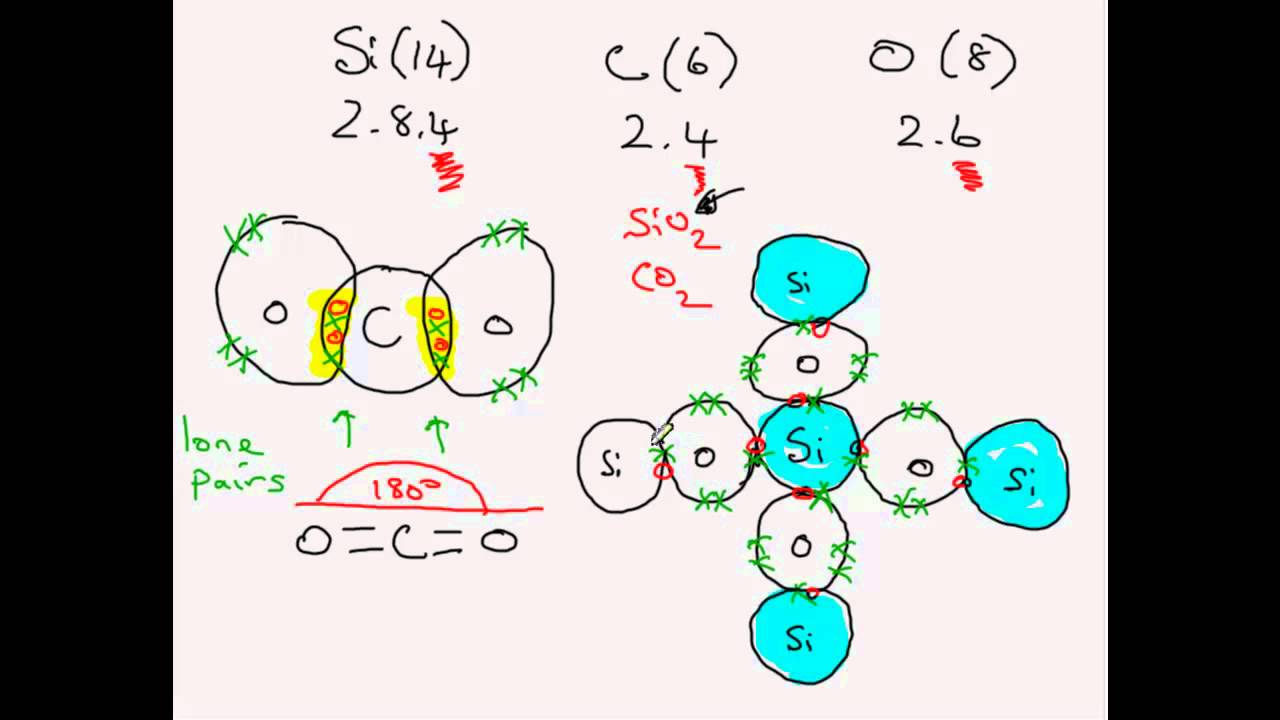
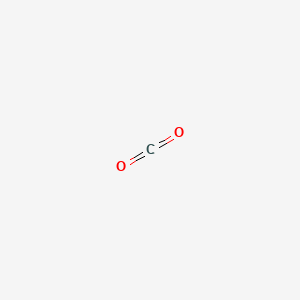
Carbon dioxide is a linear structure with two double bonds between carbon and oxygen.
Why is carbon dioxide a gas at room temperature and silicon dioxide a solid.
At room temperature co2 has only weak dispersion intermolecular forces holding the molecules together.
In math co 2 math displayed formula.
Silicon dioxide is giant molecular.
Carbon dioxide is simple molecular.
Then at that temperature molecules of co2 are tightly bonded together to be in solid form.
In case of sio2 each si atom is linked with o atom with single bond thus forming a network solid with high melting point 4 6k views.
It is a small molecule and non polar with only weak bonds between the molecules.
This is at room temperature.
Carbon dioxide is a gas at room temperature while silicon dioxide is a solid at room temperature with a melting point of 1770 c.
Carbon dioxide consists of molecules in which a single carbon atom is double bonded to two oxygen atoms.
Explain this by comparing their particles and those forces between these particles.
Thus co2 is a gas at room temperature.
Explain why silicon dioxide is a solid and carbon dioxide is a gas at room temperature.
They have a lot of energy compared to molecules in a solid.
Hence it is a gas.
Carbon dioxide is a gas at room temperature silicon iv oxide is a solid with a high.
As co2 is.
The reason why carbon dioxide is a gas and silicon dioxide is a solid is because their chemical structures are different.
Carbon is a non metal with a fairly small atomic radius close to that of oxygen.
1 educator answer silicon iv oxide has a high melting point whereas carbon dioxide is a gas.