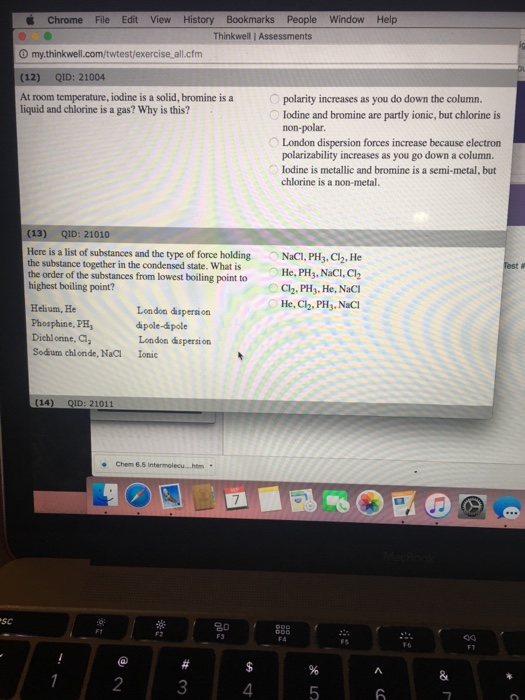
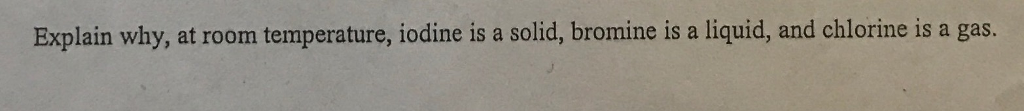At ordinary temperature and pressure chlorine is because a gas bromine is a liquid and iodine is a solid because.
Why is bromine a liquid at room temperature and iodine a solid.
In other words they are chemical elements found in group 17 of the periodic table of elements.
Bromine is a liquid at room temperature and iodine is a solid.
Iodine is further down group 7 than chlorine.
The key difference between bromine and iodine is that bromine is a liquid at room temperature while iodine is a solid at room temperature.
That s why we can observed these elements at different physical states at room temperature.
It is the third lightest halogen and is a fuming red brown liquid at room temperature that evaporates readily to form a similarly coloured gas.
As iodine has a higher atomic number than chlorine it also has more electrons in its electron shells.
Bromine and iodine are halides.
Bromine is a liquid at room temperature and iodine is a solid.
Iodine is a solid at room temperature because 500 likes.
It has a low value of electronegativity.
At room temperature is chlorine a.
Chlorine cl2 has the lowest mm thus weakest ldf while iodine i2 has the highest mm thus strongest ldf.
Bromine is a chemical element with the symbol br and atomic number 35.
Its properties are thus intermediate between those of chlorine and iodine isolated independently by two chemists carl jacob löwig in 1825 and antoine jérôme balard in 1826.
Why is iodine a solid at room temperature and chlorine a gas despite being in the same group.
The iodine that you buy in a store as a liquid has been mixed with other chemicals for easy use.
What elements form diatomic gases.
Their group number only describes the number of electrons they have in their outer shell.